
I remember Moseley very well, with whom I was on very friendly terms. But apart from the shortcomings it was a very fine lab, nice rooms, and lots of people working there-able people. So years went on without apparatus being cleaned. And of course you were not supposed to clean it. Now the microscope was fixed and then you were not supposed to touch it. But of course also a microscope to read the electroscope. And you charge the electroscope by sealing wax which you rubbed on your trousers. You have to build it yourself of cocoa boxes, gold leaf and sulfur isolation. Now the technique used in Rutherford’s lab was to fit up an electroscope. And of course everywhere you see smoke there, everywhere the smoke. Namely, Manchester is very foggy, foggy and smoky. I found Rutherford's place very busy, hard working. You need Flash Player installed to listen to this audio clip. Rutherford entered the center of the physics world. (1882–1945) because of his experimental skill, and endowed a new position in mathematical physics to round out a full physics program. Schuster had built a modern physics building, hired Hans Geiger, Ph.D. He had been named Langworthy Professor of Physics, successor to Arthur Schuster (1851–1934), who retired at age 56 to recruit Rutherford. Rutherford arrived in Manchester in the summer of 1907, months before the university's term began. AIP Emilio Segrè Visual Archives, Brittle Books Collection. Credit: From the book: The physical laboratories of the University of Manchester: a record of 25 years' work by the University of Manchester, Manchester: At the University Press, 1906. For comparison, Rutherford's generous salary was £1,600/year. At the urging of his predecessor, Arthur Schuster, over £40,000 was raised to endow the physics program. When Rutherford became professor at Manchester in 1907, he found modern labs for both teaching and research. It involved hard work and perplexity and inspiration. The story as it unfolded in Rutherford's lab at the University in Manchester revolved around real people. So this hints that perhaps the story of the discovery of the nucleus was more complicated. But can discovery be the same for a realm hidden from sight? One cannot see an atom in that sense. But what does that statement mean? Geographical discovery usually means that one sees a place for the first time. We read this in textbooks and in popular writings. One of his best-known students was Ernest Rutherford, who succeeded Thomson as Cavendish Professor of Physics.Sections ← Prev 1 2 3 4 5 6 Next → Alpha Particles and the Atom Rutherford at Manchester, 1907–1919Įrnest Rutherford discovered the nucleus of the atom in 1911. Seven of his research assistants, as well as his own son, went on to win the Nobel Prize in Physics. Thomson's greatest contribution to science to be his role as a teacher. Thomson published an important monograph in 1913 urging the use of the mass spectrograph in chemical analysis. His atomic theory helped explain atomic bonding and the structure of molecules. Thomson was closely aligned with chemists of the time.These experiments led to the development of the mass spectrograph. Thomson also investigated the nature of positively-charged particles.The son, Sir George Paget Thomson, received the Nobel Prize in Physics in 1937. In 1890, Thomson married one of his students, Rose Elisabeth Paget.So, Joseph John attended Owens College in Manchester, and then Trinity College in Cambridge, where he became a mathematical physicist. to be an engineer, but the family did not have the funds to support the apprenticeship. In 1906, Thomson demonstrated a hydrogen atom had only a single electron.Thomson discovered the natural radioactivity of potassium in 1905.Thomson's master's work, Treatise on the motion of vortex rings, provides a mathematical description of William Thomson's vortex theory of atoms. 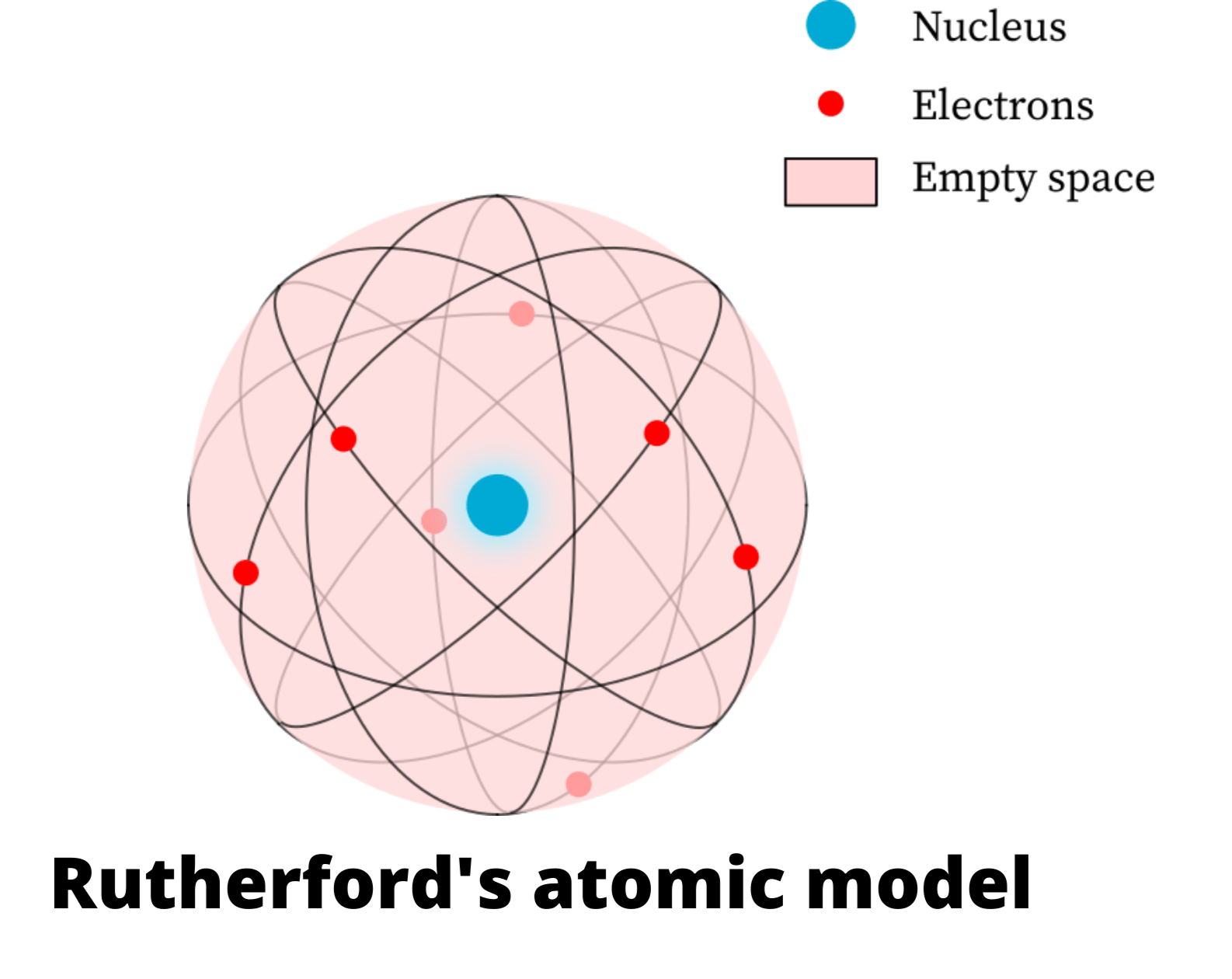
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
